Marie Skłodowska-Curie ETN MgSafe
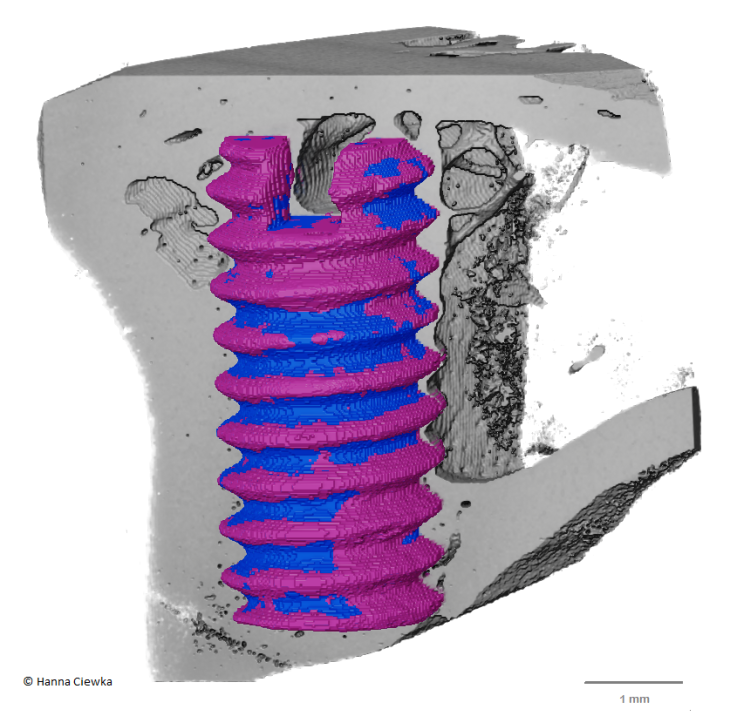
Volume rendering of biodegradable Mg-based screw in bone obtained from high-resolution micro computed tomography. The purple colour represents the degraded metal that has formed on the surface of the remaining metal (blue).
MgSafe is a European Training Network within the framework of Horizon 2020 Marie Skłodowska-Curie Action (MSCA) 2018.
Within this action, 15 Early Stage Researchers (ESRs) address the optimisation of imaging technologies for biodegradable magnesium implants.
Fractures are typically treated with non‐degradable metal implants, which commonly require surgical removal after complete bone healing. From the health care and patients’ point of view, degradable implants provide a viable, cost effective and patient friendly alternative. In 2013, the first degradable metal implant made from a Mg‐alloy (compression screw of partner SYNTELLIX) was CE certified and has be implanted into several 100 patients so far.
Monitoring implant performance and degradation with the existing imaging techniques is a challenge
The ESRs of MgSafe will push the imaging modalities towards their limits to monitor the degradation processes of emerging Mg implants optimally and non‐invasively in animal models with high spatial and temporal resolution.
The results of MgSafe will substantially increase the level of safety for patients currently treated with Mg‐based implants and will boost the further development of imaging modalities also on a clinical level.
MgSafe will educate a new generation of young researchers needed for the development of high‐tech medical devices.
How MgSafe will improve patient safety and what the project is all about:
This project has received funding from the European Union’s Horizon 2020 research and innovation programme under the Marie Skłodowska-Curie grant agreement No 811226.